
Within a group (family) of elements, atoms form ions of a certain charge. The charge that an atom acquires when it becomes an ion is related to the structure of the periodic table. Generally, on a periodic table, all of the elements in a single vertical column will have the same number of valence electrons. They gain two electrons and have a -2 charge. Students should be familiar with the following terms: valence electrons. The oxygen family elements have 6 valence electrons. Students will discover trends of ion charges with relation to the periodic table. The halogens (Cl, F, Br) all have 7 valence electrons and will gain one electron. Valence electrons are the electrons in the outermost shell, or energy level, of an atom. Chromium is a transition element and should lose its s electrons and then its d electrons when forming a cation.\): Predicting Ionic Charges. A modified form of Coulomb's Law is written below, where e is the charge of an electron, Z e f f is the effective nuclear charge experienced by that electron, and r is the radius (distance of the electron from the nucleus). The members of the aluminum family lose three electrons giving them a charge of +3. 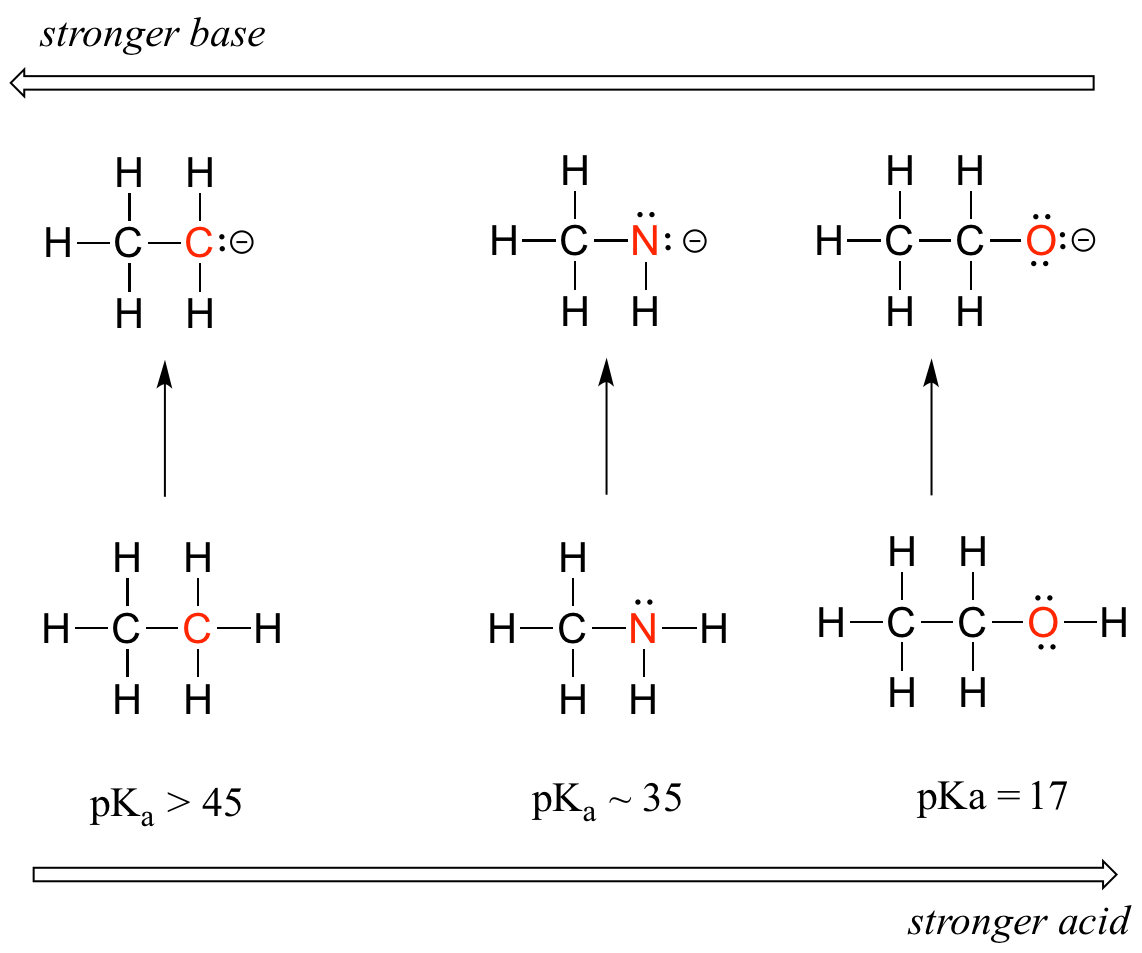
Zinc is a member of group 12, so it should have a charge of 2+, and thus loses only the two electrons in its s orbital. For the p-block elements, electrons are removed from the p orbitals and then from the s orbital. Many of the periodic properties of atoms depend on electron configuration in particular, the valence electrons and their level of. An atom of an element is most stable when its outer electron shell is completely filled or half-filled. One can use the periodic table to predict whether an. (b) A sodium cation (Na+) has lost an electron, so it has one more proton (11) than electrons (10), giving it an overall positive charge, signified by a superscripted plus sign. 
The charge on an atom is related to its valence electrons or oxidation state. 1: (a) A sodium atom (Na) has equal numbers of protons and electrons (11) and is uncharged. You can use this chart to predict whether or not an atom can bond with another atom. It can be approximated by the equation: Zeff Z S, where Z is the atomic number and S is the number of shielding electrons. For the transition metals, electrons are removed from the s orbital first and then from the d orbital. This is a chart of the most common charges for atoms of the chemical elements. Next, remove electrons from the highest energy orbital. Write the electron configurations of these cations.įirst, write the electron configuration for the neutral atoms: Two of these essential trace elements, chromium and zinc, are required as Cr 3 + and Zn 2 +. 
They are called “essential” because they are required for healthy bodily functions, “trace” because they are required only in small amounts, and “elements” in spite of the fact that they are really ions. Metals in Groups 1A, 2A and 3A will form ions with 1+, 2+ and 3+ charges, respectively. In fact, the group that a main-group element is associated with in the periodic table will dictate the valence (or charge) of its corresponding ion. There are at least 14 elements categorized as “essential trace elements” for the human body. Elements in Group 2A can each lose two electrons to form 2+ ions and achieve a noble gas configuration. The relationship between valence electrons and charge of an ion is EXTREMELY IMPORTANT. \): Determining the Electronic Structures of Cations
0 Comments
Leave a Reply.AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
